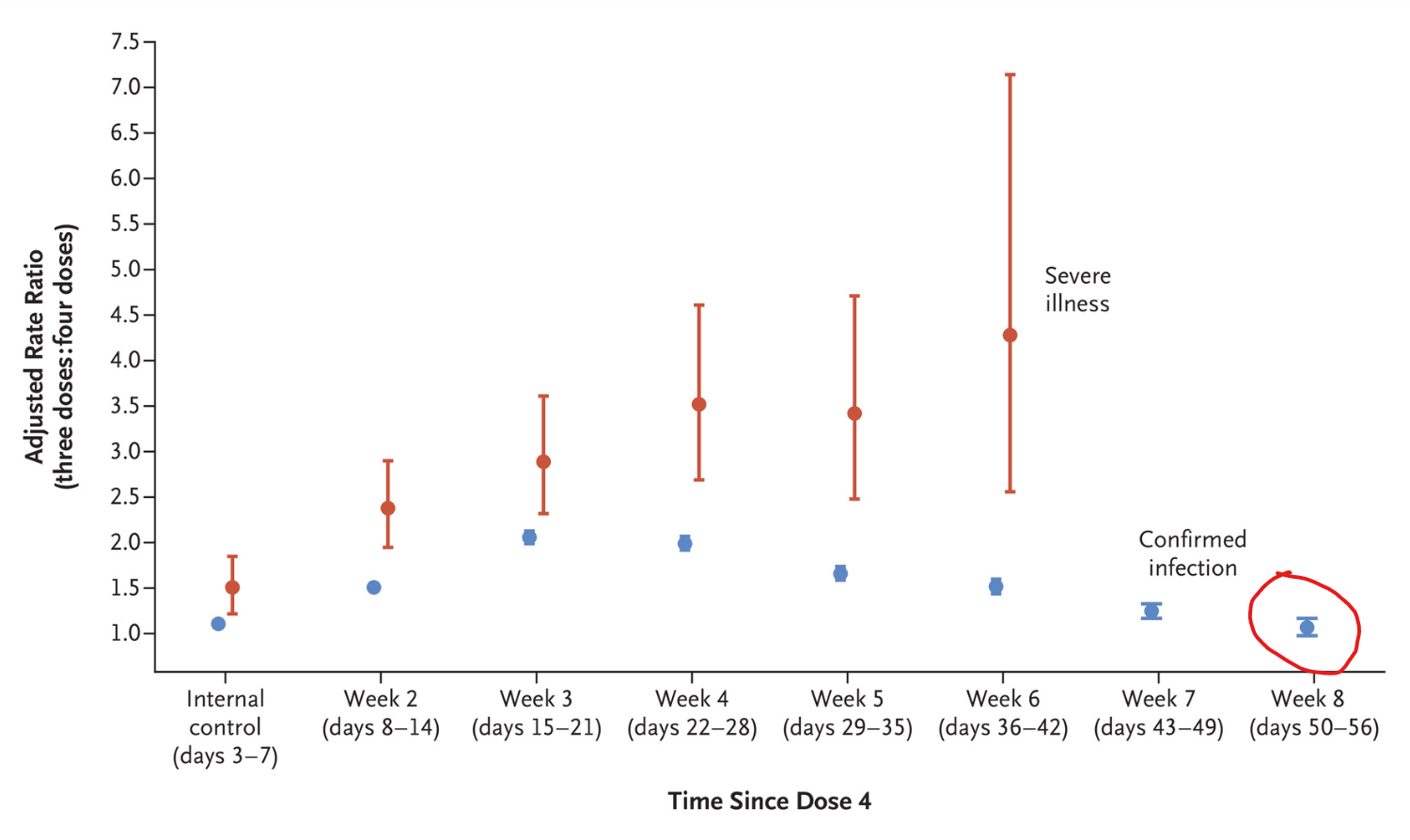
A population-wide study from Israel published in the New England Journal of Medicine has found that a fourth dose of the Pfizer Covid vaccine ceases to have any efficacy against infection within just eight weeks (see chart above). Here is the abstract.
BACKGROUND
On January 2nd 2022, Israel began administering a fourth dose of BNT162b2 [Pfizer] vaccine to persons 60 years of age or older. Data are needed regarding the effect of the fourth dose on rates of confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and of severe coronavirus disease 2019 (COVID-19).
METHODS
Using the Israeli Ministry of Health database, we extracted data on 1,252,331 persons who were 60 years of age or older and eligible for the fourth dose during a period in which the B.1.1.529 (Omicron) variant of SARS-CoV-2 was predominant (January 10th through March 2nd 2022). We estimated the rate of confirmed infection and severe COVID-19 as a function of time starting at eight days after receipt of a fourth dose (four-dose groups) as compared with that among persons who had received only three doses (three-dose group) and among persons who had received a fourth dose three to seven days earlier (internal control group). For the estimation of rates, we used quasi-Poisson regression with adjustment for age, sex, demographic group, and calendar day.
RESULTS
The number of cases of severe COVID-19 per 100,000 person-days (unadjusted rate) was 1.5 in the aggregated four-dose groups, 3.9 in the three-dose group, and 4.2 in the internal control group. In the quasi-Poisson analysis, the adjusted rate of severe COVID-19 in the fourth week after receipt of the fourth dose was lower than that in the three-dose group by a factor of 3.5 (95% confidence interval [CI], 2.7 to 4.6) and was lower than that in the internal control group by a factor of 2.3 (95% CI, 1.7 to 3.3). Protection against severe illness did not wane during the six weeks after receipt of the fourth dose. The number of cases of confirmed infection per 100,000 person-days (unadjusted rate) was 177 in the aggregated four-dose groups, 361 in the three-dose group, and 388 in the internal control group. In the quasi-Poisson analysis, the adjusted rate of confirmed infection in the fourth week after receipt of the fourth dose was lower than that in the three-dose group by a factor of 2.0 (95% CI, 1.9 to 2.1) and was lower than that in the internal control group by a factor of 1.8 (95% CI, 1.7 to 1.9). However, this protection waned in later weeks.
CONCLUSIONS
Rates of confirmed SARS-CoV-2 infection and severe COVID-19 were lower after a fourth dose of BNT162b2 vaccine than after only three doses. Protection against confirmed infection appeared short-lived, whereas protection against severe illness did not wane during the study period.
While protection against severe disease did not appear to decrease during the six week follow-up, note that the fact that severe Covid illness was occurring in the triple-dosed at over three times the rate of the quadruple-dosed suggests that the protection of three doses against severe illness had already significantly declined. This is confirmed by other studies (e.g. here and here) and noted by the authors themselves.
Read the study here. My write-up of an earlier version can be found here.











To join in with the discussion please make a donation to The Daily Sceptic.
Profanity and abuse will be removed and may lead to a permanent ban.