The latest Public Health England (PHE) weekly vaccine surveillance report appeared on Thursday (another one is due tomorrow).
I wasn’t holding out high hopes for this instalment, after digging down into the method used the previous week and finding it didn’t control adequately for background incidence (so confounded vaccine effectiveness with natural decline) and applied such brutal adjustments it could turn a 4% effectiveness into a 73% one. It lived down to expectations.
The authors appear still to be working out what exactly their reports are for as each of the three to date presents different information in a slightly different way. While the first was largely a summary of evidence published elsewhere, the second served more as a showcase for PHE’s latest study and focused on effectiveness against symptomatic disease in the over-65s (90%, supposedly). This week, the over-65s are gone and the focus is on effectiveness against the Indian variant. This makes reading the reports quite confusing, as each doesn’t feel so much like an update to the previous one as a stand-alone report on a separate topic.
In between Report 2 (for week 20) and Report 3, a separate PHE study was published on the Indian Delta variant which found the effectiveness of full vaccination against symptomatic Covid reduced from 93% to 88% with Pfizer and 66% to 60% with AstraZeneca when the Indian Delta variant was involved versus the British Alpha variant.
As I noted last week, this 66% effectiveness of AstraZeneca (against the British Alpha variant) was a long way below the 90% in the over-65s claimed in Report 2. It’s probably a sign of how low my opinion is of PHE vaccine studies that I was pleasantly surprised to see this discrepancy actually make an impact on the data presented in Report 3, and you can see below in the AstraZeneca two-doses box (top right) week 20’s “85-90%” has become “65% to 90%” in week 21. There is still no explanation from PHE, however, as to why one of their studies finds AstraZeneca 90% effective against symptomatic infection in the over-65s while another finds it is just 66% effective in all ages. Is this not the kind of discrepancy that should occasion comment from the authors?
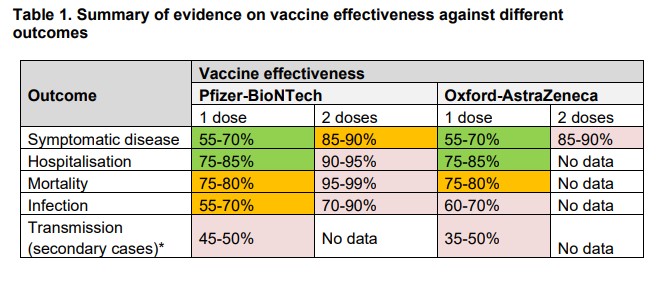
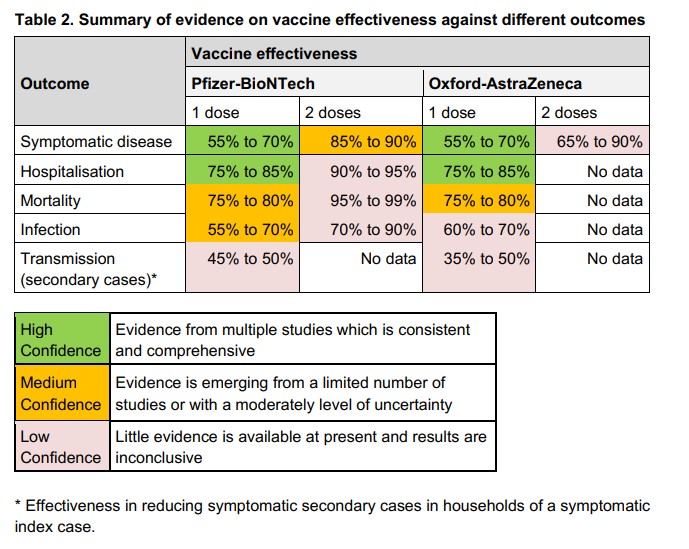
Another moment where full transparency seems to be lacking is in the section about the Indian Delta variant, where rather than presenting the figures for each vaccine separately the authors combine the data for the two vaccines to give 81% overall effectiveness against the new variant.
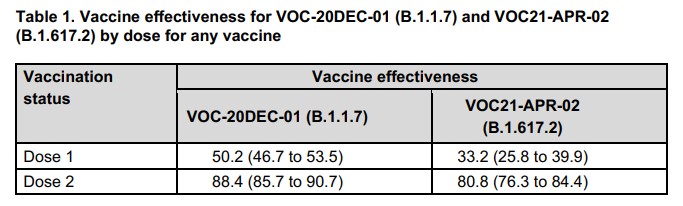
This seems a strange decision given that this breaks down to 88% for Pfizer and 60% for AstraZeneca, making Pfizer almost 50% more effective than AstraZeneca against the Indian Delta variant. Was this done to spare AstraZeneca’s blushes? Certainly there’s no sign of PHE drawing attention to this disappointing AZ data. We might wonder though: is part of what is driving the recent fear-mongering from SAGE scientists a worry that the Oxford “workhorse” vaccine is not up to the job, particularly in the most vulnerable?
Another oddity is that the report states that “by May 23rd 2021, the overall vaccine uptake in England for dose 1 was 51.3% and 31.4% for dose 2”. Yet the Government dashboard shows 72% of the adult population had one dose and 42% had two doses by that date. The reason? The report is counting children in the unvaccinated population, but without noting this fact.
Overall my feeling is that these reports have been of limited usefulness (so far), and manage to confuse as often as they clarify. What we really need from PHE are studies which control properly for background incidence so we can get a proper picture of how effective the vaccines actually are, including in the frail elderly where many of the deaths are concentrated. We are also waiting for a proper analysis of the post-vaccination infection spike, rather than half-baked attempts to explain it away, and are long overdue studies that include analysis of safety and not just efficacy. Will PHE step up to the plate?












To join in with the discussion please make a donation to The Daily Sceptic.
Profanity and abuse will be removed and may lead to a permanent ban.