In a recent article in the BMJ, a whistle-blower exposed serious problems she had observed first-hand in the Pfizer vaccine trial in Texas.
A regional director who was employed at the research organisation Ventavia Research Group has told the BMJ that the company falsified data, unblinded patients, employed inadequately trained vaccinators, and was slow to follow up on adverse events reported in Pfizer’s pivotal phase III trial. Staff who conducted quality control checks were overwhelmed by the volume of problems they were finding. After repeatedly notifying Ventavia of these problems, the regional director, Brook Jackson, emailed a complaint to the US Food and Drug Administration (FDA). Ventavia fired her later the same day. Jackson has provided the BMJ with dozens of internal company documents, photos, audio recordings, and emails.
Another Ventavia employee said of the data the company generated for the Pfizer trial: “I don’t think it was good clean data. It’s a crazy mess.”
The six-month trial results for the Pfizer vaccine have now been published in the New England Journal of Medicine. These findings, the researchers note, “contributed to the full approval of BNT162b2 [the Pfizer vaccine] in the United States”. A close inspection of the study, however, reveals a number of problems that raise serious questions about the reliability of its findings, as well as about the safety of the vaccine.
Here is the graph of cumulative incidence for the two trial arms, vaccine and placebo, over the six months of the study period, showing how the symptomatic Covid PCR-positives added up following receipt of the first dose.
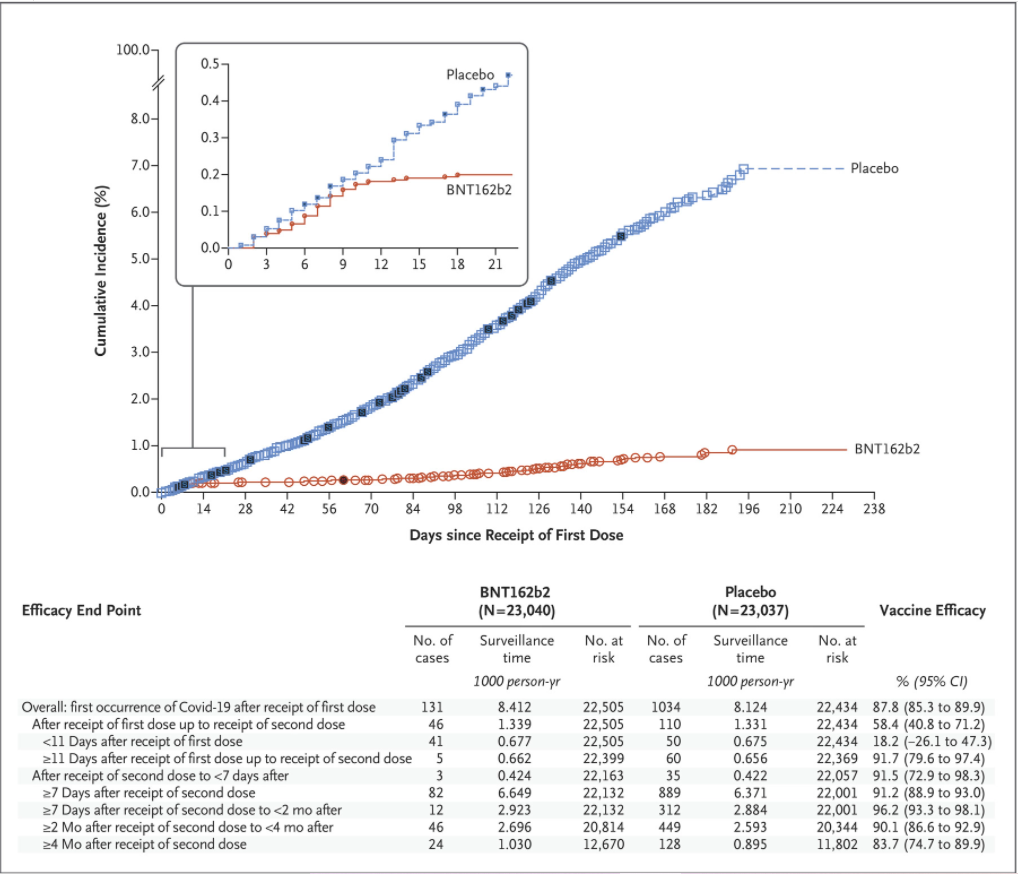
The first curious feature is that vaccine efficacy abruptly kicks in on day 12 after the first dose. Prior to that it’s near zero (18.2%), as expected, but then all of a sudden incidence in the vaccinated arm grinds almost to a halt and the vaccine efficacy leaps to 91.7% and stays there. Second doses were given at 21 days, but there is no sign of (or room for) an uplift in efficacy at that point. This high efficacy of a first dose at day 12 is in conflict with the results of a real-world mass observational study in Israel covering a similar time period (December 20th to February 1st), which found vaccine effectiveness against symptomatic Covid for days 14-20 after the first dose to be just 57%.
The trial study authors note that vaccine efficacy drops to 83.7% after 4-6 months, a fall of around 3% per month. Where, though, is the sharper decline seen in other studies, such as those from Sweden and Qatar? Is it because the study period ends on March 13th 2021, so pre-dates not only Delta but also Alpha in the countries where the trial was being carried out (mainly USA, Brazil and Argentina)? It’s worth noting that during the observational study period Israel did have a high prevalence of Alpha. If this is part of the reason for the discrepancy, it calls into question how applicable the findings are to the present context where Delta dominates.
Unfortunately, the trial can no longer help to answer these questions as, the authors explain, “most participants who initially received placebo have now been immunised with BNT162b2, ending the placebo-controlled period of the trial”.
Another question is why the severe cases (marked in black on the chart above) are not evenly distributed. Why do they appear in clumps with gaps, when the other symptomatic infections increase evenly? Also, there is only one severe case in the vaccine arm and 30 in the placebo arm (giving a reported efficacy against severe disease of 96.7%). Yet three of the severe cases in the placebo arm occurred in the first 12 days after the first dose, before the vaccine is supposed to start working and when there is no difference in the numbers of non-severe cases. Why then are there no severe cases in the vaccine arm in that period? Why, also, is the ratio of deaths in the two arms one-to-two (vaccine to placebo) but the ratio of severe cases one-to-30?
Some of the figures for people who dropped out (see below) are also odd.
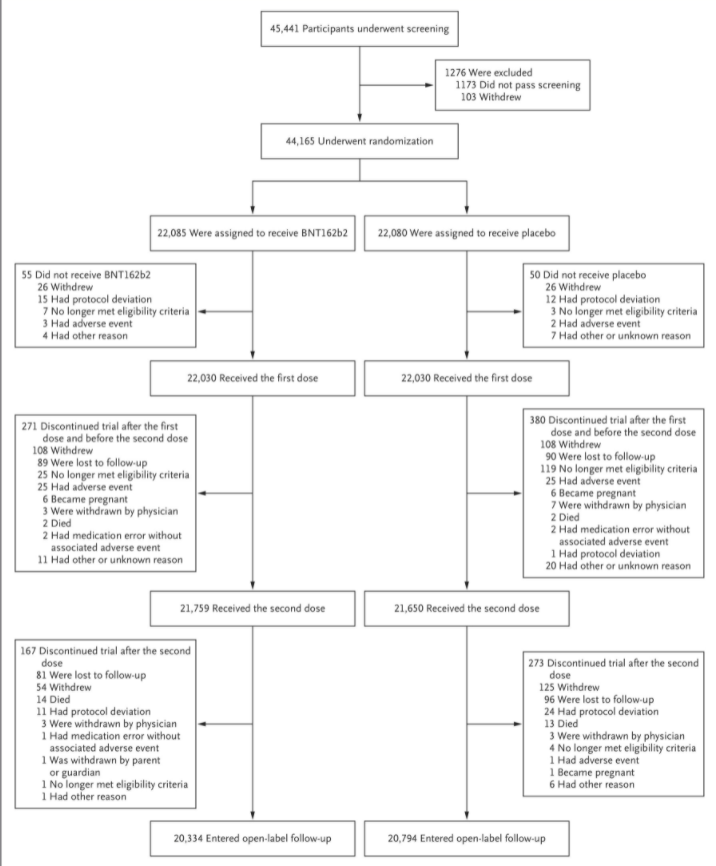
Note that in each arm, prior to one dose, exactly 26 people withdrew, and after one dose, exactly 108 people withdrew and 25 had an adverse event (odd considering the placebo was saline). Six became pregnant and two died in each arm, while 89 (vaccine) and 90 (placebo) were “lost to follow up”. A professor of statistics wryly observed to me it was “as if they have gone to some trouble to show how remarkably well randomised the trial was”.
Someone sent me this handy graphic showing all the deaths recorded in the study.
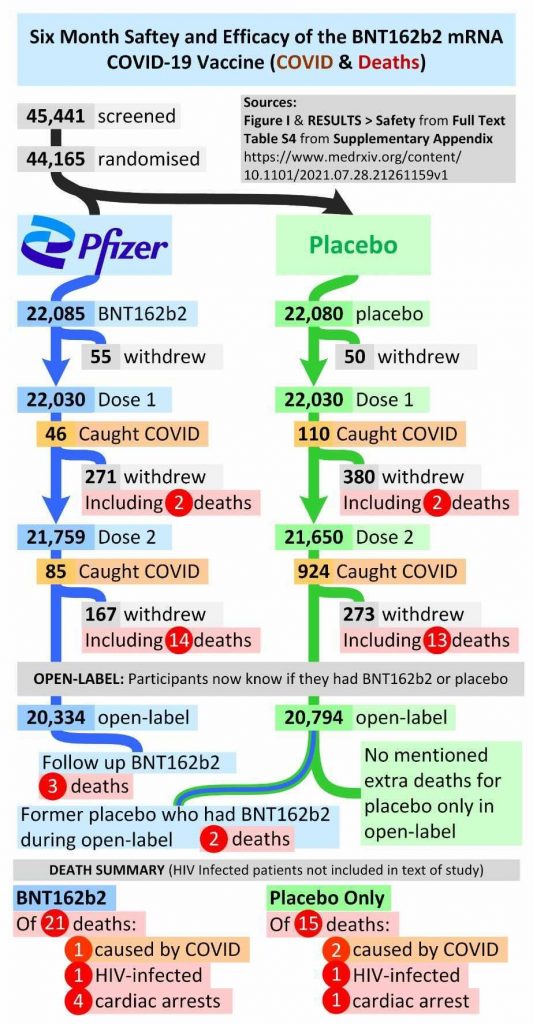
Notably there are 21 deaths from all causes among the vaccinated and 15 among the unvaccinated – though if we exclude the five from the period after the unblinding when most of the placebo group received the vaccine (and no more deaths in the placebo group were recorded) it’s 16 vs 15. Alex Berenson has spotted a report where Pfizer state it was actually 21 vs 17 by the end of the blinded study period on March 13th, raising questions of why there seems to be ambiguity on this important data point. Either way, there are more deaths from all causes in the vaccine arm than the placebo arm, and significantly, perhaps, one of the big differences by cause is there are four versus one heart attacks. Despite this, the researchers claim: “Causes of death were balanced between BNT162b2 and placebo groups.”
Inevitably, though: “None of these deaths were considered to be related to BNT162b2 by the investigators.” Also: “No cases of myocarditis were noted.”
The data on natural immunity is confusing. The authors state that: “Participants with a history of COVID-19 were excluded, although evidence of current or previous SARS-CoV-2 infection on laboratory testing of trial-obtained samples was not an exclusion criterion.” This suggests they excluded those who had had symptomatic Covid before, but not those who’d tested positive asymptomatically. However, then they state: “Nine cases of COVID-19 were observed among participants with previous serologically defined natural infection: two cases were observed among the vaccine recipients and seven among the placebo recipients.”
How can there be a significant number of people in the trial with SARS-CoV-2 antibodies if they excluded anyone with a history of COVID-19 – assuming that you don’t get antibodies from an infection so mild it produces no symptoms? This needs to be cleared up, not least because the authors claim their data “support the current practice of immunising without screening for evidence of previous infection”.
With so many questions and concerns about the trial and its data, it’s hard to know how it can be trusted, despite supposedly being the gold standard of evidence.
That’s certainly the feeling of Surya Arby, a consultant in France, whose BMJ ‘rapid response‘ to the whistle-blower report remarks: “It’s hard to understand how we can trust the safety data provided by Pfizer.” Surya notes that “the official package insert approved by the FDA for Comirnaty [Pfizer] states that acute allergic reactions (including anaphylaxis) have been reported only in post-marketing surveillance (including EUA)”. However, “in the real world, the observed rate of acute allergic reactions is close to 2% (1.95% [1.79%-2.13%]) and the observed rate of anaphylaxis is close to 1/3,700 for mRNA COVID-19 vaccines (Pfizer 0.027% [0.011%-0.056%])”.
Such a rate is “impossible to miss,” he says, “in a cohort of 21,700 vaccinated individuals in a clinical trial”.
Indeed, yet ‘miss’ it they did.











To join in with the discussion please make a donation to The Daily Sceptic.
Profanity and abuse will be removed and may lead to a permanent ban.